About Ensho Therapeutics
Ensho means inflammation, flame, or glow in Japanese and reflects our deep desire to deliver breakthrough oral therapies to patients with inflammatory diseases.
We are advancing a potential best-in-class oral, small molecule α4β7 inhibitor, known as NSHO-101, that could be transformational for treating patients suffering from inflammatory bowel disease (IBD). α4β7 inhibition is a mechanism that has already been shown to be safe and effective by a commercially available biologic, but unmet needs remain and no approved oral α4β7 inhibitors currently exist.
For the millions of patients who suffer from IBD, we hope to extinguish their inflammation and provide relief from this difficult-to-treat disease.
Quiet the Fire Within
Chronic inflammation is a hallmark of IBD, ultimately damaging the gastrointestinal tract.
IBD is characterized by debilitating symptoms, including abdominal pain, diarrhea, and weight loss that greatly impact quality of life. Because IBD frequently begins in early adulthood, often between the late teens and mid-30s - the years most associated with education, career development, and family formation - the disease can carry a disproportionate burden on quality of life and economic productivity.
The most widely prescribed medications to address the symptoms of IBD are biologics, which require cumbersome infusions and / or injections and can lead to non-adherence.
For these young adults with high demands on career, family, and social obligations, an effective and safe oral therapy could provide a more convenient and promising treatment option: one that better fits their lifestyles.

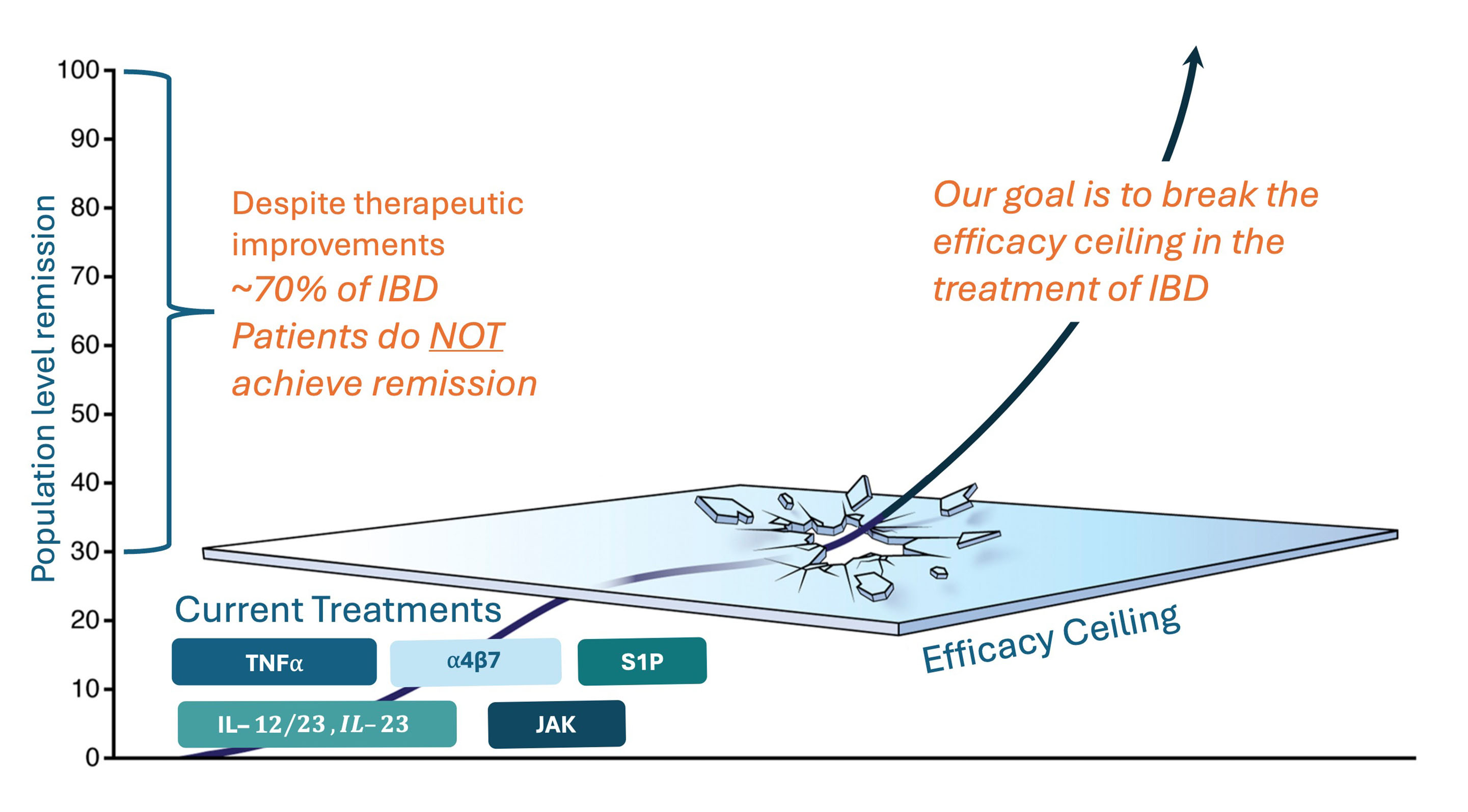
Breaking the Efficacy
Ceiling in the Treatment of IBD
The approval of biologics, including an α4β7 blocking antibody, has improved outcomes, but the majority of patients do not achieve or maintain remission – commonly referred to as the “efficacy ceiling.” Many patients experience persistent symptoms and / or develop intolerance to, or experience side effects from, their treatment regimens. There is a push to move beyond current therapies and provide more transformational treatments, including combination therapy, to potentially improve the overall outcomes for patients with IBD. Combining the right orthogonal mechanisms of action has the potential of additive efficacy without overlapping side effects. This approach is currently being studied in a number of ongoing Phase 2 trials.
Ensho’s goals are to provide an oral alternative to biologic treatments and to break through the efficacy ceiling in combination with other oral mechanisms of action. Our drug candidate, NSHO-101, is designed as a potential best-in-class once-daily, oral therapy that has the potential to be the preferred oral alternative to biologic therapy, with additional potential to act as the anchor of choice for oral fixed-dose combination therapy.
New transformational treatments are needed for IBD
Adapted from Raine et al., Gastroenterology (2022).
α4β7 inhibition is one of the most validated immune-trafficking targets in IBD
α4β7 is a cell surface receptor that helps regulate the migration of immune cells to the intestine and plays a key role in controlling inflammatory responses. It binds to mucosal addressin cell adhesion molecule-1 (MAdCAM-1), which is expressed on the luminal side of endothelial cells and is upregulated in response to inflammation. This interaction facilitates the transport of leukocytes and recruitment of effector lymphocytes to the gut mucosa in IBD. Because MAdCAM-1 is found primarily in the gut, this localized action provides immune cell inhibition where it is most relevant, potentially minimizing side effects and preserving overall immune cell function.
Decades of research validate α4β7 inhibition as an anti-inflammatory mechanism in IBD. This research has been further substantiated by the approval of a safe and efficacious commercially available antibody, vedolizumab. Yet, there are no approved orally administered α4β7 inhibitors. That’s where NSHO-101, our oral, selective, small molecule inhibitor of α4β7 can play a role.
NSHO-101: A potential best-in-class oral α4β7 inhibitor
NSHO-101 is a highly potent, selective, small molecule α4β7 inhibitor with a potential best-in-class profile optimized for convenient once daily oral administration – a profile designed to enable combination therapy with other mechanisms of action.
NSHO-101 was acquired from EA Pharma, a subsidiary of Eisai, Ltd. and Ajinomoto, based on robust Phase 1 data that demonstrated greater than 90% α4β7 receptor occupancy at trough with QD dosing
We substantially improved NSHO-101’s tablet formulation to enable convenient and combinable once daily dosing, which is currently being confirmed in a Phase 1 healthy volunteer study. Planning for Phase 2 clinical development is underway.
Leadership
Our leadership team includes a seasoned team of executives with decades of experience in the discovery, development, and launch of inflammation & immunology agents as well as the management of biotechnology companies in the inflammatory disease and IBD space including oral a4b7 small molecules.
Gerhard Hagn, PharmD
Chief Executive Officer
Danielle Fry, JD
Chief Operating Officer and Chief Legal Officer
Tory Trippe
Vice President, Head of Finance and Corporate Development
Matthew R. Warr, PhD
Senior Vice President, Research & Head of Biology
Andy Whitney, PhD
Chief Scientific Officer
 Gerhard Hagn, PharmD
Gerhard Hagn, PharmD
Chief Executive Officer
Gerhard Hagn, PharmD, is Chief Executive Officer at Ensho. He brings to Ensho over 25 years of industry experience. He most recently served as Chief Commercial Officer at Tourmaline Bio, a biopharmaceutical company focused on treatments for Immunology and Inflammation (I&I) diseases, leading commercial and business development which culminated in the $1.4 billion acquisition of Tourmaline Bio by Novartis. Prior to that, he built Gilead's inflammation business unit including their oral a4b7, led the $3 billion liver franchise, and was responsible for advancing commercial efforts for 20 established products. Prior to Gilead, Mr. Hagn spent 20 years at Pfizer Inc., where he most recently acted as the I&I business unit lead responsible for reimbursement, pricing, and evidence generation of launches and in-line business in Rheumatology, Gastroenterology, Dermatology, and for assets advancing into Phase 3 clinical trials.
Gerhard holds an Apotheker degree (PharmD) from the Ludwig-Maximilians-Universität in Munich, Germany.
 Danielle Fry, JD
Danielle Fry, JD
Chief Operating Officer and Chief Legal Officer
Danielle is the Chief Operating Officer and Chief Legal Officer at Ensho, where she brings extensive experience in business development, alliance management, and clinical development. Before joining Ensho, Danielle played a critical role at Eisai Inc. where she supported business development efforts and managed key strategic alliances, including collaborations in clinical development.
Prior to her time at Eisai, Danielle was part of the Reed Smith Life Sciences Transactions group, where she worked on mergers and acquisitions, licensing agreements, and other high-impact transactions within the life sciences sector.
Danielle began her career as a scientist at Schering-Plough, where she conducted research in HIV and Hepatitis C, contributing to the development of key therapeutic programs. Her hands-on experience in scientific research provided her with a deep understanding of the complexities of drug development and clinical trials.
Danielle holds a B.S. in biology from The College of New Jersey and following her time at Schering-Plough, obtained a law degree (J.D.) at Seton Hall Law School, where she honed her expertise in life sciences transactions and regulatory affairs. This unique combination of scientific and legal knowledge has been pivotal in her roles across the pharmaceutical and biotech industries.
 Tory Trippe
Tory Trippe
Vice President, Head of Finance and Corporate Development
Tory Trippe is the Vice President, Head of Finance and Corporate Development at Ensho, where she brings over 10 years of experience spanning management consulting, corporate strategy, and finance across the biopharmaceutical industry.
Most recently, Tory served as Senior Director of Finance and Corporate Development at Tourmaline Bio, a biopharmaceutical company focused on treatments for I&I diseases. She contributed to the company's growth and strategic trajectory, which culminated in the $1.4 billion acquisition of Tourmaline by Novartis. Prior to Tourmaline, Tory held strategic finance roles at Roivant Sciences and Alexion Pharmaceuticals, supporting strategic planning and business development initiatives across rare and specialty disease portfolios. Tory began her career as a management consultant at Bain and Company, advising clients across multiple industries.
Tory holds a B.S. in Biology and Applied Economics and Management from Cornell University.
 Matthew R. Warr, PhD
Matthew R. Warr, PhD
Senior Vice President, Research & Head of Biology
Matthew Warr is the Senior Vice President, Research & Head of Biology of Ensho. Matthew brings to Ensho more than 12 years of experience of industry research experience where he advanced multiple small molecule I&I programs from discovery into clinical development.
Prior to Ensho, Matthew served as the Head of Inflammation and Immunology Research, Drug Discovery at Kallyope, a leading gut-brain axis privately held biotechnology company. At Kallyope, he led a team of I&I researchers, directing efforts for a pipeline of small molecules being developed for inflammatory gastrointestinal diseases, including IBD, food allergy, and celiac disease. Matthew began his career at Gilead Sciences in the Biology group where he held leadership roles on multiple inflammation and oncology research projects.
Matthew holds a BScH from Queen’s University in Biochemistry and a Ph.D. in Biochemistry from McGill University.
 Andy Whitney, PhD
Andy Whitney, PhD
Chief Scientific Officer
Andy Whitney is the Chief Scientific Officer of Ensho. Andy brings extensive experience in autoimmune/inflammatory, oncology and rare diseases drug discovery and development. He has a proven track record building and leading dynamic and successful research teams in both small and large companies. Andy has extensive project leadership experience, with multiple programs advancing from target selection through clinical development and has contributed to the successful development of two U.S. FDA-approved therapeutics.
Andy’s 25 years of experience includes leading the preclinical drug development as Senior Vice President, Head of Preclinical Development and Translational Science at Applied Molecular Transport, and prior to that, leading preclinical biology efforts for multiple rare disease projects as Vice President, Biology at BridgeBio. Previously, Andy was a Director in Gilead’s inflammation and oncology research group, where he held site leadership roles and led research efforts for multiple small molecule programs in inflammation and oncology. He was previously one of the earliest staff at CGI Pharmaceuticals, Inc., where he led biology, bioinformatics and project teams until its acquisition by Gilead.
Andy holds an A.B. from Harvard College and a Ph.D. in Cell Biology from Yale and was a National Science Foundation postdoctoral fellow at the University of Geneva, Switzerland.
